Performance of Acrylic
We were asked to look at the performance of acrylic and the force required to break an acrylic window, fitted into a caravan which had been in service since 2001. Unfortunately sample of the glazing was not made available for inspection and subsequent physical analysis.
Impact Solutions are a UKAS accredited ISO 17025 lab and are a UK’s notified body under the construction products regulations. Therefore, we have extensive experience in failure analysis across a wide range of materials and industries from the oil & gas, construction, infrastructure and packaging and are capable of determining factors which could influence the performance of acrylic.
It is accepted that the use of acrylic materials in motorhome windows and roofing has been a very common practice over the last decades. Compared with the mineral glass windows, acrylics are cheaper to make, as well as they yield better mechanical properties. Their biggest advantage though is their lower density, which makes them much lighter than glass with an immediate effect on the material transport and fuel costs. But, acrylics, like most of the commodity plastics have a main disadvantage against the glass windows; they are susceptible to prolonged thermal/UV exposure and/or chemical attack (organic solvents etc) and could eventually become more brittle. These two factors have been the main cause for a series of acrylic failures in the past. It is reasonable to assume that a caravan in service from 2001 would have been exposed to significant UV exposure (sunlight), and a wide range of chemical exposure from different household chemicals or solvents and will also be subjected to scratches and chips from its time in service. All of these factors are likely to have significantly reduced the impact performance of the window.
UV exposure
Acrylics are vulnerable to thermal, UV radiation and chemical attack which originates from their fundamental molecular structure. Unlike conventional mineral glasses, acrylics are organic materials based mainly on the polymethyl-methacrylate, PMMA polymer. UV radiation from sun exposure can cause a photochemical effect within the polymer structure, which can lead to degradation to the material. The stronger the UV radiation or the longer the exposure to UV the more profound the degradation will be. Absorbed UV energy can excite the photons in a polymer-plastics, which then in turn create free radicals. While many pure plastics cannot absorb UV radiation, the presence of catalyst residues and other impurities (examples: oxygen, sodium) even at trace amounts, will often act as free radical receptors. Those free radicals can eventually cause breaks in the covalent polymer bonds causing degradation and render the material more susceptible to catastrophic brittle failures. The degradation of the polymer would also give this distinctive yellow finish on the polymer surface evident with the naked eye.
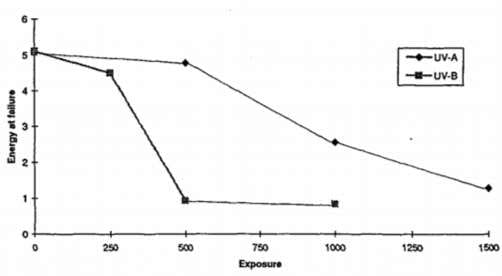
The effect of the UV exposure on the mechanical performance of polymers has been of the primary focus on both development scientists and engineers and numerous studies-reports have been published. In the particular case of the impact performance of acrylic glazing (PMMA) polymer upon exposure to UV, this has been investigated by S M Haliwell, “Weathering of plastics glazing materials”, Loughborough University, 1996. In this study, PMMA windows have been exposed to accelerated weathering machines (Q-UV) and tested for their impact performance using the falling weight method following the ISO 6603-1:1985 method. The graph shown in figure 1 below, presents the impact resistance of the PMMA against the accelerated weathering time under UV-A and UV-B conditions. It is evident from the graph that the energy at break is reduced to almost 50% after 1000hours of exposure to UV-A which corresponds to 1 year of actual exposure to sun.
As precautionary measures, acrylics are usually filled with UV additives to protect against weathering. But, it is unpractical and not cost-effective to fill the acrylics with too much UV additives, and as result the level of additives depends on the application and on the target cost of the finished product. It is very likely that the acrylic glazings are UV protected, but their protection is limited to the level of exposure, use and age of the glazing. Common UV additives are being consumed into water molecules during time.
In a number of other high value external products we typically see 10 years of UV protection, so assuming the window has been in service since 2001, it is very likely to have been significantly affected by UV exposure, which in turn dramatically reduces its impact properties.
Thermal degradation
Plastics show reduced mechanical performance when exposed to prolonged times at elevated temperatures (higher than room temperatures) OR at low temperatures close to freezing. This is typical exposure of an external product and we all know how hot a car can get in the summer sun. In the first case, molecules soften and this may allow the protective additives leach out the plastics or accelerates the thermal degradation. In the second, low temperature, case the molecules of the plastics are more “stiff” i.e. in a frozen state, reducing dramatically their impact performance.
Chemical attack
Acrylic could fail prematurely or become brittle following an attack or contamination of specific chemicals which are for household applications. Acetone (found in nail varnish remover), for example even in tiny quantities attacks and dissolves the PMMA molecules leaving micro cracks behind significantly weakening the overall material.
Conclusion
With a product that has been in service since 2001, it is reasonable to assume that the prolonged exposure to UV radiation, environment and chemical attack will have significantly weakened the impact properties of the acrylic rendering it more brittle causing catastrophic fractures even at low impact energies.
If you have any enquiries, please don’t hesitate to get in touch via phone on +44 (0) 1324 489 182 or email us at info@impact-solutions.co.uk.
- SM Haliwell, Weathering of plastics glazing materials, 1996
- UV degradation Mechanisms, http://www.drb-mattech.co.uk/uv%20degradation.html
- Toshimi Hirata et al., Thermal and oxidative degradation of PMMA: weight loss, Macromolecules, 1985, volume 18
- Feld, M. S. et. al. “The Physics of Karate,” Scientific American, pp. 150-158, April 1979
Impact Solutions
+44 1324 489 182
Website
Email







