WHP Succeeds with Med-tech Projects
Wittmann Battenfeld UK client White Horse Plastics continues to invest in its medical and healthcare markets by offering value-added design and manufacturing projects to OEMs and specifiers worldwide.
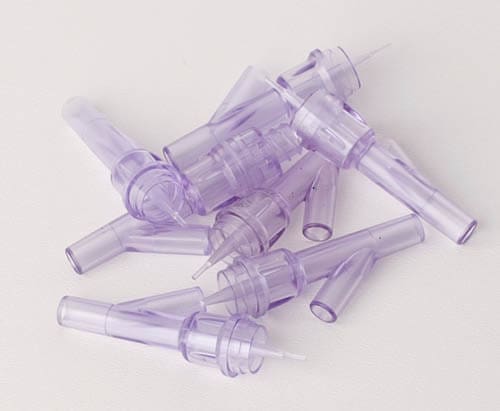
The Oxfordshire-based injection moulding innovator is extending its reach into the med-tech sector with the tooling and manufacture of luer connectors. WHP’s ISO 13485 qualification gained last year now enables the company to pro-actively market its well-recognised manufacturing skills to the worldwide medical-manufacturing supply chain.
Renowned innovation consultancy PDD, headquartered in London was tasked by its international client to invent and develop four design variants of a luer device– three straight and the other a Y shaped device. Drawings and CAD data were supplied to WHP together with a materials specification. The focus was finalising the design for multi cavity tooling suitable for future high volume production.
In partnership with both PDD and its client, WHP helped assess the novel and challenging design for manufacture and assembly (DfM&A), before building fully hardened pilot tooling and moulding the parts in a medical grade of polycarbonate for final validation studies. The company also assembled and packaged the finished components before shipping.
Dave Eyles, WHP project manager says that ‘tight tolerances were demanded for these intricate medical parts. We moulded, assembled and ultra-sonically welded (two mouldings and a silicone bellows insert) in-house within a Class 10,000 clean room for the customer’.
WHP tackles the most challenging aspects of tight tolerance technical injection moulding and sub-assembly manufacture. The company’s recent moves into the medical sector are supported by its long experience in cost effective moulding for the automotive industries, bringing sophisticated sourcing and purchasing strategies to the med-tech clients.
The WHP business was founded in 1973 and the company services range from development, design, and in house tool-manufacture through to lean-manufactured serial production. In addition to the new ISO 13485 accreditation, the key standard for suppliers of med-tech manufacturing services, WHP also holds ISO9001, ISO 14001, and TS 16949, the exacting standard specifically for the automotive industry.
WHP material experience ranges from standard polymers, nylons, acetals, polycarbonates, ABS and TPEs etc through to the more exotic engineering polymers.
Wittmann Battenfeld
+44 (0) 1933 275777
Website
Email






